Many students and learners get confused about the difference between endothermic and exothermic reactions because both involve energy changes, but in opposite ways.
You’ll often see these terms in chemistry classes, exams, and even real life processes like cooking or burning fuel.
In simple words, the difference between endothermic and exothermic comes down to energy absorption vs energy release.
One takes in heat from the surroundings, while the other gives heat out. This small difference completely changes how the reaction behaves.
If you’ve ever wondered “what is an endothermic reaction?”, “how does an exothermic reaction work?”, or “why does temperature change?”,
this guide will explain everything clearly with examples, tables, and real world applications. By the end, you won’t need another article.
🟢 Quick Answer: Difference Between Endothermic and Exothermic
- Endothermic reaction absorbs heat from surroundings
- Exothermic reaction releases heat into surroundings
👉 Example:
- Melting ice = endothermic
- Burning wood = exothermic
🟢 Definition of Endothermic and Exothermic
- Endothermic: A chemical reaction that absorbs energy (heat) from its surroundings.
- Exothermic: A chemical reaction that releases energy (heat) into its surroundings.
🟢 Core Energy Concept (Very Important)
\Delta H = H_{products} H_{reactants}
👉 If ΔH (enthalpy change) is positive, the reaction is endothermic.
👉 If ΔH is negative, the reaction is exothermic.
This formula is the backbone of understanding the difference between endothermic and exothermic.
🟢 Pronunciation (US & UK)
- Endothermic: /ˌɛn.doʊˈθɜːr.mɪk/ (US), /ˌɛn.dəʊˈθɜː.mɪk/ (UK)
- Exothermic: /ˌɛk.soʊˈθɜːr.mɪk/ (US), /ˌɛk.səʊˈθɜː.mɪk/ (UK)
👉 Even though the words sound technical, the concept is actually simple once you connect it to heat flow.
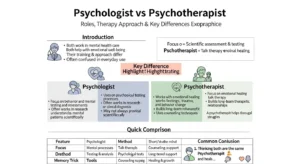
🟢 Comparison Table: Endothermic vs Exothermic
| Feature | Endothermic | Exothermic | Similarity |
| Energy Flow | Absorbs heat | Releases heat | Both involve energy change |
| Temperature Effect | Surroundings cool down | Surroundings heat up | Affect environment |
| Enthalpy (ΔH) | Positive | Negative | Measured in energy units |
| Example | Photosynthesis | Combustion | Chemical reactions |
| Energy Source | External | Internal release | Energy transformation |
| Real life Feel | Feels cold | Feels hot | Observable effects |
🟢 Key Differences Explained Between Endothermic and Exothermic
1. Energy Direction
- Endothermic → energy goes into the system
- Exothermic → energy comes out of the system
👉 Example: Ice melting vs fire burning
2. Temperature Change
- Endothermic → surroundings get cooler
- Exothermic → surroundings get warmer
3. Bond Energy Concept
- Endothermic reactions require energy to break bonds
- Exothermic reactions release energy when bonds form
4. Enthalpy Sign (ΔH)
- Positive ΔH → Endothermic
- Negative ΔH → Exothermic
5. Real Life Feeling
- Endothermic feels cold
- Exothermic feels hot
6. Energy Storage vs Release
- Endothermic stores energy
- Exothermic releases stored energy
🟢 Why Do Endothermic and Exothermic Reactions Exist?
This concept exists because of how chemical bonds work. In chemistry, energy is required to break bonds and released when new bonds form.
- If breaking bonds needs more energy → endothermic
- If forming bonds releases more energy → exothermic
This balance determines the nature of every chemical reaction.
🟢 How Endothermic Reactions Work
In an endothermic reaction:
- Energy is absorbed from surroundings
- Molecules gain energy
- Reaction feels cold
Examples:
- Ice melting
- Evaporation of water
- Photosynthesis
👉 In photosynthesis, plants absorb sunlight to produce food.
🟢 How Exothermic Reactions Work
In an exothermic reaction:
- Energy is released into surroundings
- Molecules lose energy
- Reaction feels hot
Examples:
- Burning fuel
- Respiration in humans
- Combustion
👉 Combustion releases heat and light energy.
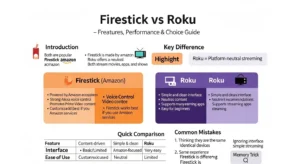
🟢 Difference and Similarity Table (Detailed)
| Feature | Endothermic | Exothermic | Similarity |
| Heat Transfer | Absorbed | Released | Energy involved |
| System Energy | Increases | Decreases | Energy change |
| Surroundings | Cool down | Heat up | Observable |
| Reaction Type | Energy intake | Energy output | Chemical reactions |
| Example | Melting | Burning | Real life processes |
This table clearly shows the difference and similarity between endothermic and exothermic for quick understanding.
🟢 Common Mistakes with Endothermic and Exothermic

❌ 1. Mixing heat direction
✔ Remember: Endo = “in”, Exo = “out”
❌ 2. Ignoring temperature effect
✔ Cold = endothermic, Hot = exothermic
❌ 3. Confusing ΔH signs
✔ Positive = endothermic, Negative = exothermic
❌ 4. Thinking all reactions release heat
✔ Some absorb energy
❌ 5. Memorizing without understanding
✔ Focus on energy flow logic
🟢 Real Life Examples of Endothermic and Exothermic
Daily Life
- Ice packs → endothermic
- Fire → exothermic
Biological Systems
- Photosynthesis → endothermic
- Respiration → exothermic
Industrial Use
- Chemical manufacturing
- Fuel combustion
Cooking
- Baking absorbs heat
- Frying releases heat
🟢 When to Use Each Concept
Use Endothermic Concept when:
- Studying cooling processes
- Understanding energy absorption
- Analyzing plant biology
Use Exothermic Concept when:
- Studying heat generation
- Understanding fuels and combustion
- Industrial reactions
🟢 Why People Get Confused
The confusion in the difference between endothermic and exothermic happens because:
- Similar terminology
- Opposite meanings
- Complex scientific language
- Lack of practical examples
🟢 How Search Engines Understand This Topic
Search engines like Google use NLP to detect educational intent:
- Queries like “difference between endothermic and exothermic” trigger explanation based content
- Content with formulas, examples, and tables ranks higher
That’s why structured, example rich content performs best.
🟢 Expert Insight
In real scientific practice, understanding endothermic and exothermic reactions is essential in fields like chemistry, engineering, and environmental science.
Professionals analyze energy changes to design efficient reactions. For example:
- Engineers optimize fuel combustion (exothermic)
- Scientists study energy storage systems (endothermic)
Mastering this concept builds a strong foundation for advanced chemistry.
🟢 FAQ:
1. What is the main difference?
Endothermic absorbs heat; exothermic releases heat.
2. Which reaction feels cold?
Endothermic reactions.
3. Which reaction releases energy?
Exothermic reactions.
4. Is photosynthesis endothermic?
Yes.
5. Is burning fuel exothermic?
Yes.
6. What does ΔH mean?
Energy change in a reaction.
7. Why is this important?
It explains energy flow in chemistry.
🟢 Conclusion
Understanding the difference between endothermic and exothermic is one of the most important concepts in chemistry.
These reactions explain how energy moves in the world around us from plants absorbing sunlight to fuels burning and releasing heat.
In simple terms, endothermic reactions take in energy, while exothermic reactions release it. This difference affects temperature, reaction behavior, and real world applications.
By remembering the core idea Endo = energy in, Exo = energy out you can easily identify and understand any reaction. With examples, formulas, and real life connections, this concept becomes clear and practical.
Once you master this, you’ll find chemistry much easier and more interesting.
Discover More Articals!
Difference Between Affect and Effect (Simple Words + Examples)
Difference Between Mono and Strep: Symptoms, Causes & Treatment

James Whitmore. Labdiff.com is my english grammar comparision website. I explore the history and evolution of English words. I write detailed comparisons explaining why spellings change over time and how British and American English diverged. My articles add depth and credibility to grammar comparison topics.